Next-generation sequencing (NGS) clinical laboratories rely on different types of software to meet their business and laboratory needs. Leading laboratories often work with experienced software consultants to make sure that each piece of software is verified and validated, and set up using the best software engineering practices, integrations, and automations.
Key Clinical Laboratory Systems Fulfilled by Software
The software systems that NGS clinical labs employ vary widely from one company to the next, depending on the lab’s core business, but there are standard functions that these labs require. Rather than seeing each piece of lab software as a standalone platform, we view the laboratory flow as a series of integrated systems, or in software jargon, a “stack.”
Commonly, labs need some or all of the following in their stack:
- A system of record for the patient journey through the health system.
- Electronic medical records (EMR) or electronic health records (EHR). These are often used or integrated into hospital labs and independent labs with hospital customers to keep track of patient health information and enable clinicians to order tests efficiently.
- A system of record for the patient’s specimen through the laboratory that enables sample submission, status information, accessioning, and result delivery.
- Laboratory information systems (LIS). In hospitals, this system is the main lab management tool for managing tests and patient data coming from the EMR.
- Order management system. In independent laboratories, this system provides a clinical web portal and other interfaces for receiving orders and delivering results. Labs often use SalesForce, Orchard, or a fully custom solution.
- A system of record for the sample, its derivatives, the quantitative results, and the associated workflows.
- Laboratory information management systems (LIMS). These are utilized to manage laboratory operations by guiding lab analysts through each step of an assay workflow and recording and archiving all pertinent data at each step. They are primarily responsible for tracking wet lab analysis but can be integrated with a variety of other systems.
- A system for translating raw data from wet lab analysis into diagnostic reports.
- Analysis software. This is predominantly the realm of bioinformatics, alignment, and big data processing in the NGS context.
- Interpretation software. Allows professional interpretation of genetic variants, in the context of an NGS lab. This may or may not include cross-reference functionality to databases like PubMed, but will always include the ability to draft and sign a diagnostic report, which then moves into the result delivery mechanisms referenced above.
- A system for reporting metrics on laboratory operations.
- Operational reporting software enables data-driven insights into the day-to-day operations of the lab.
- A system to manage inventory of laboratory consumables and reagents.
- Inventory, freezer, and consumable management systems, typically integrated with the LIMS.
- Systems to manage regulatory compliance submissions and accreditations, as well as the quality programs associated with them.
- Regulatory and quality software systems, including document control systems. These are typically layered across all the systems mentioned above.
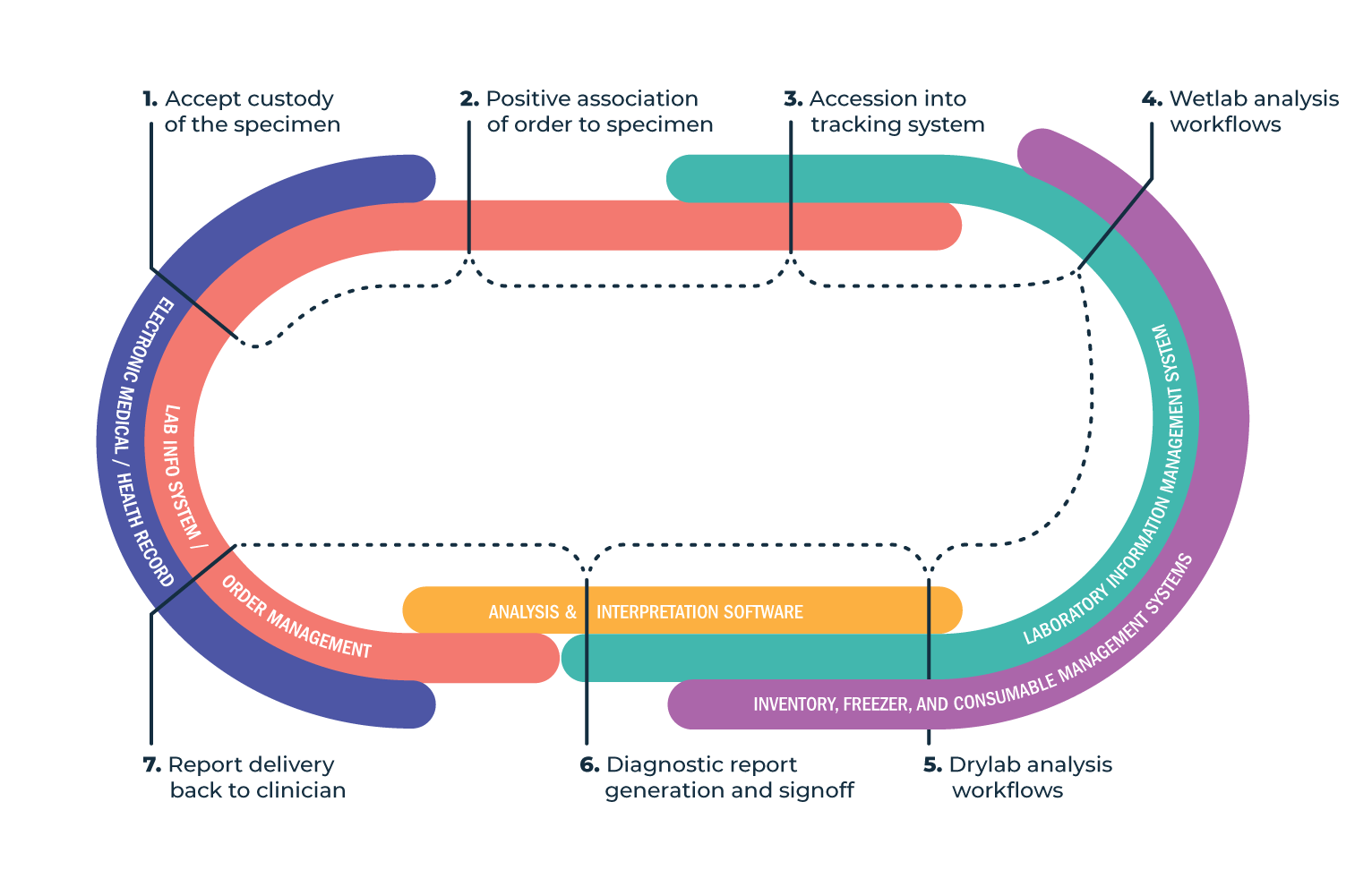
The role of software in a specimen’s typical life cycle in a clinical laboratory
To use a juggling analogy, that’s a lot of balls to keep in the air. And all it takes is a small slip to spoil the performance. Issues can arise if the software is out-of-date, components aren’t deeply integrated, or the system lacks automated processes. Labs with poorly constructed software also run the risk of not meeting regulatory requirements—they might have limited traceability or even use incorrect calculations. Or they could suffer time-to-market delays when releasing new products, tests, or assays.
4 Reasons to Get Expert Help with Your Laboratory Software
Ensuring that your NGS clinical lab has a software stack that runs efficiently and enables you to scale throughput is key to gaining a competitive advantage. Specifically, here are four reasons why you should invest in optimizing your software stack:
1. Integrated systems save time and reduce human error
By integrating the various components of a software stack, you can streamline processes and mitigate duplicated effort. With the addition of appropriate automations, your lab will free up staff time for more complex tasks and reduce the potential for error—manual processes are, without fail, more prone to human error.
2. Good software practices lead to software that can be easily modified and upgraded
Well-designed and integrated software gives your lab more agility when changes need to be made. This enables the lab and software teams to concentrate on the lab’s overarching business strategy rather than the nuts and bolts of how new or updated software components will fit into the system.
3. Well-documented and repeatable processes help you meet regulatory requirements
Consultants can help make certification, accreditation, or regulatory approvals quicker and simpler. A consultant with a thorough knowledge of the granularities of industry regulatory programs will ensure your software stack and complex workflows meet your traceability and reporting goals, for any accreditation program you’re pursuing.
4. An efficient software stack means your lab is ready to scale
A key competitive advantage for labs today is the ability to scale, which means higher volumes can be processed and with greater efficiency. An expert is able to evaluate if your lab software is up for the challenge, undertaking performance modeling and testing to confirm your systems are designed for scalability and peak efficiency.
There’s No One-Size-Fits-All Approach
NGS clinical labs require a complex mix of software solutions. Unfortunately, there’s no one-size-fits-all approach or a single off-the-shelf software product that will give you all the tools you need—which is why so many labs end up adopting a piecemeal approach to their software stack.
At Semaphore, we think there’s a better way. We follow engineering best practices and rigorous processes to help you craft and validate your lab’s software stack, ensure the components are all up-to-date, integrate the pieces that make sense, and add automations that will help you be more efficient—and, ultimately, more competitive.
Contact us to discuss how your software stack can do a better job supporting your business goals.

